Bioinorganic, Bioorganic, Biophysical Chemistry
- B.A. Harvard College (1994)
- Research Fellow, Johannes Gutenberg Universitaet, Mainz, DE (1994-1995)
- Ph.D. California Institute of Technology (2000)
- Caltech Herbert Newby McCoy Award (2000)
- Helen Hay Whitney Postdoctoral Fellow, Biophysics, Caltech (2000-2002)
- Camille and Henry Dreyfus New Faculty Award (2003)
- National Science Foundation CAREER Award (2005)
- Camille and Henry Dreyfus Teacher-Scholar Award (2007)
Our lab is developing chemical and biophysical tools to study and manipulate complex biological systems. Projects span many areas of synthetic organic, inorganic, and biophysical chemistry; molecular, cell, and developmental biology; and bioengineering. We are particularly interested in developing new technologies for biomolecular imaging and the fabrication of functional bio-nanomaterials.
Hyperpolarized 129Xe Biosensors for Early Cancer Detection
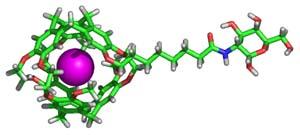
Molecular imaging technologies hold great promise for early cancer diagnosis and intervention. Our goal is to develop new reagents that extend the capabilities of magnetic resonance imaging (MRI) for monitoring multiple cancer markers simultaneously in vivo. 129Xe has found increasing use for biological imaging applications, due to its biological compatibility (xenon is an anesthetic at high concentrations), hyperpolarizability (this enhances signals 1,000-fold), and high affinity for organic cages such as cryptophanes. The chemical shift of 129Xe varies by a remarkable 200 ppm, depending on its molecular environment: Thus, a 129Xe atom encapsulated inside a cryptophane is a sensitive reporter of perturbations outside the cage. Based on this principle, our lab is generating new biosensors that will identify biomarkers associated with cancers of the breast, lungs, brain, and pancreas. The long-range goal of this project is to use MRI to detect aberrant proteins that cause cancer in humans, years before the formation of a tumor.
Ferritin Templates for Nanoparticle Synthesis and Assembly
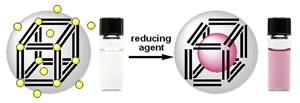
The goal of this project is to use ferritin proteins as templates for synthesizing and assembling inorganic nanoparticles with nanometer precision. Ferritins contain 24 four-helix bundle subunits that self-assemble to create a large central cavity. We have made water-stable, 10-12-nm gold and silver nanoparticles inside ferritin (gray sphere). Particles are fully characterized using facilities at the UPenn Laboratory for Research on the Structure of Matter (LRSM). We are functionalizing the surface of these ferritin-metal nanoparticles for sensing and nano/biomaterials applications. We are also performing computational protein design, in collaboration with the Saven lab, to mutate residues inside the ferritin cavity to enhance their metal-binding properties. Methods for organizing ferritin metal nanoparticles in 2- and 3-dimensions are being developed, in order to build very small conducting circuits.
Laser-Activated Chemical Biology: Controlling Genes with Light
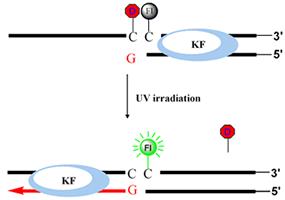
The goal of this project is to develop methods for turning genes "on" and "off" with light inside neurons and developing zebrafish embryos with high spatial and temporal control. As a first step, we have developed methods for incorporating a photoactive blocking group in the middle of a DNA or RNA oligonucleotide. In one application, we modulated primer extension by DNA polymerase (KF) using UV light. Photoactivation was monitored using a fluorescent reporter. We are now developing methods to control protein translation by the ribosome using similarly caged RNA. Blocking groups mask the messenger RNA start codon, and are designed to prevent translation until photocleavage. We will control complex gradients of proteins involved in cell signaling during zebrafish development and wound healing, using a state-of-the-art UV confocal microscope in the lab.
- Chemistry 101, "General Chemistry"
- Chemistry 559, "Biomolecular Imaging"
- Chemistry 567, “Bioinorganic Chemistry”
X. Tang, J. Swaminathin, A.M. Gewirtz, I.J. Dmochowski, Regulating gene expression in human leukemia cells using light-activated oligodeoxynucleotides, Nucl. Acids Res. (36) 559-569, 2008.
J.A. Aaron, J.M. Chambers, K.M. Jude, L. Di Costanzo, I.J. Dmochowski, D.W. Christianson, Structure of a 129Xe-cryptophane biosensor complexed with human carbonic anhydrase II, J. Am. Chem. Soc. (130) 6942-6943, 2008.
G.K. Seward, Q. Wei, I.J. Dmochowski, Peptide-mediated cellular uptake of cryptophane, Bioconjug. Chem. (19) 2129-2135, 2008.
J.L. Richards, X. Tang, A. Turetsky, I.J. Dmochowski, RNA bandages for photomodulating in vitro protein synthesis, Bioorg. Med. Chem. Lett. (18) 6255-6258, 2008.
C. Butts, J. Swift, S.-G. Kang, L. Di Costanzo, D.W. Christianson, J.G. Saven, I.J. Dmochowski, Directing noble metal ion chemistry within a designed ferritin protein, Biochemistry (47) 12729-12739, 2008.
J.L. Chambers, P.A. Hill, J.A. Aaron, Z. Han, D.W. Christianson, N.N. Kuzma, I.J. Dmochowski, Cryptophane xenon-129 nuclear magnetic resonance biosensors targeting human carbonic anhydrase, J. Am. Chem. Soc. (131) 563-569, 2009.
P.A. Hill, Q. Wei, T. Troxler, I.J. Dmochowski, Substituent effects on xenon binding affinity and solution behavior of water-soluble cryptophanes, J. Am. Chem. Soc. (131) 3069-3077, 2009.
G.P. Robbins, M. Jimbo, J. Swift, M.J. Therien, D.A. Hammer, I.J. Dmochowski, Photo-initiated destruction of composite porphyrin-protein polymersomes, J. Am. Chem. Soc., (131) 3872-3874, 2009.
J. Swift, C. Butts, J. Cheung-Lau, V. Yerubandi, I.J. Dmochowski, Efficient self-assembly of Archaeoglobus fulgidus ferritin around metallic cores, Langmuir, (25) 5219-5225, 2009.
C.A. Butts, J. Xi, G. Brannigan, M.L. Klein, R.G. Eckenhoff, I.J. Dmochowski, Identification of a fluorescent general anesthetic, 1-aminoanthracene, Proc. Natl. Acad. Sci. U.S.A. (106) 6501-6506, 2009.
I.J. Dmochowski, Xenon out of its shell, Nature Chemistry, ‘In Your Element’ invited feature article, vol. 1, 250, June 2009.
O. Taratula, I.J. Dmochowski, Functionalized 129Xe contrast agents for magnetic resonance imaging, Curr. Opin. Chem. Biol. (14) 97-104, 2010.
J.L. Richards, G.K. Seward, Y. Huang, I.J. Dmochowski, Turning DNAzymes on and off with light, ChemBioChem (11) 320-324, 2010.
J. Lampe, Z. Liao, I.J. Dmochowski, P.S. Ayyaswamy, D.M. Eckmann, Imaging macromolecular interactions at an interface, Langmuir (26) 2452-2459, 2010.

