Biological, Chemical Physics and Physical Chemistry
- B.S. University of Michigan (1964)
- Ph.D. University of Michigan (1968)
- USPHS Career Development Award (1971-76)
- Guest Biophysicist, Brookhaven National Laboratory (1973-present)
- Chairman, Department of Chemistry (1983-1986)
- Director, Biostructures Participating Research Team, National Synchrotron Light Source, Brookhaven National Laboratory (1985-1994)
- Executive Committee, Complex Materials Collaborative Access Team, Advanced Photon Source, Argonne National Laboratory (1994-present)
- Scientific Advisory Committee & Advisory Board, Spallation Neutron Source, Oak Ridge National Laboratory (1997-2006)
- Executive Committee, Cold Neutrons in Biology and Technology Team, National Institutes of Standards and Technology (1998-2006)
- Director, Complex Materials Collaborative Access Team, Advanced Photon Source, Argonne National Laboratory (2001-present)
Our research program falls into two areas, namely nano-scale materials science and fundamental biophysics (or biophysical chemistry). The materials science effort is directed toward the development of novel electro-optical devices, both single particle and 2-D to 3-D ensemble based, utilizing the unique microscopic properties of designed cofactor-artificial peptide complexes. The cofactors are based primarily on extended conjugated chromophores designed to exhibit light-induced electric charge transfer over large nano-scale distances and possess minimal HOMO-LUMO bandgaps. The highly stable, artificial α-helical peptides are based on n-helix bundle structural motifs designed to vectorially incorporate the cofactor within the core of the bundle and to order the assembly of the peptide-cofactor complexes at the liquid-gas, solid-liquid or solid-gas interface. Ensembles of these complexes have potential for both photovoltaic devices applications relevant to solar energy conversion and non-linear optical device applications relevant to broad-band communications. The structures and properties of both the isolated complexes and the ensembles thereof are determined by cutting-edge techniques, including molecular dynamics simulation, synchrotron x-ray and cold neutron scattering, and polarized CW and transient spectroscopies. The biophysics effort is directed toward understanding the mechanism of volatile general anesthetic action on membrane ion channels. To better access the physical chemistry of the anesthetic-protein interaction, we are utilizing artificial membrane ion channels based on an amphiphilic 4-helix bundle motif, the hydrophilic domain designed to possess the anesthetic binding cavity and the hydrophobic domain designed as a membrane-spanning cation channel. The same techniques mentioned above are utilized to probe the nature of the anesthetic-peptide interaction and its effect on the conformation of the ion channel domain. Future work will also be directed toward understanding the mechanism of electro-mechanical coupling in the substantially more complex, natural voltage-gated ion channels under the control of the applied transmembrane electrochemical potential. These studies will employ cutting-edge time-resolved synchrotron x-ray and cold neutron scattering techniques coupled with molecular dynamics simulation, and will lead to the investigation of the effects of anesthetic binding on this mechanism. All of the work mentioned above involves both extensive collaborations with other faculty in the Department, at Penn, and elsewhere, as well as experimental work at the National Laboratories on a regular basis.
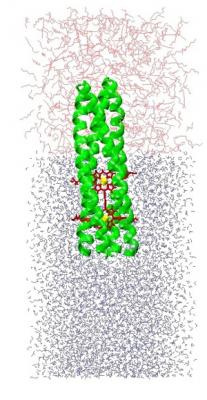
Figure Legend #1: An instantaneous configuration from a molecular dynamics simulation of the structure of an extended conjugated chromophore (a butadiyne-bridged Zn-porphyrin dimer: red/yellow) incorporated into the core of the hydrophilic domain of an amphiphilic 4-helix bundle peptide (green ribbon representation) and vectorially-oriented at the water-octane (gray-pink) interface. The time-averaged structure of the chromophore-peptide complex within a monolayer ensemble at the interface has been determined experimentally. The designed coiled-coil structure of the 4-helix bundle induces a twist in the chromophore that is key to optimizing its non-linear optical polarizability.
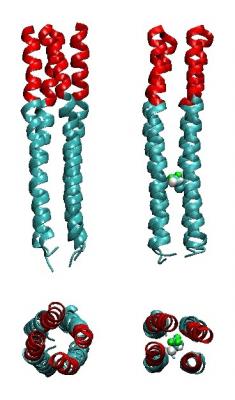
Figure Legend #2: Instantaneous configurations from molecular dynamics simulations of the structures of a computationally designed, model anesthetic-binding membrane ion channel vectorially-oriented at the water-octane interface (water/octane not shown; see Figure 1). The hydrophilic domain (blue ribbon representation) of the amphiphilic 4-helix bundle peptide contains the anesthetic-binding cavity with the volatile anesthetic halothane (CPK representation) in the cavity shown on the right-side, with the ion channel hydrophobic domain (red ribbon representation). The helices are relatively straight and un-coiled in the side-on view (upper), as more readily seen in the end-on view shown below. Removal of the anesthetic from the cavity is seen to induce a coiled-coil structure extending from the cavity into the ion channel hydrophobic domain, shown in the side-on and end-on views on the left side. Importantly, this anesthetic-dependent conformational change depends upon the amino acid composition of the cavity. Experimental verification of these predictions from the computational design and molecular dynamics simulations are underway.
Gupta, S., Dura, J., Freites, A. Tobias, D. and Blasie, J.K. Structural characterization of the voltage sensor domain and voltage-gated K+-channel vectorially-oriented within a single bilayer membrane at the solid/vapor and solid/liquid interfaces via neutron interferometry. Submitted.
Koo, J., Park, J., Tronin, A., Zhang, R., Krishnan, V., Strzalka, J., Kuzmenko, I., Therien, M.J. and Blasie, J.K. Acentric 2-D Ensembles of D-br-A Electron-Transfer Chromophores via Vectorial Orientation within Amphiphilic n-Helix Bundle Peptides for Photovoltaic Device Applications. Langmuir, 28: (6), pp 3227-3238
Gupta, S., Liu, J., Strzalka, J. and Blasie, J.K. (2011) Profile Structures of the VSD and KvAP Channel Vectorially-Oriented in Single Membranes at Solid-Vapor or Solid-Liquid Interfaces via X-ray Reflectivity. Phys. Rev. E. 84(3): 031911-1-15.
Korendovych, I., Senes, A., Kim, Y.H., Lear, J., Fry, H.C., Therien, M.J., Blasie, J.K., Walker, F.A. and DeGrado, W.F. (2010) De Novo Design and Molecular Assembly of a Transmembrane Diporphyrin-Binding Protein Complex. J. Am. Chem. Soc. Comm. 132: 15516-15518.
http://pubs.acs.org/Volume=132
Gonella, G., Dai, H.-L., Fry, H. C., Therien, M. J., Krishnan, V., Tronin, A. and Blasie, J.K. (2010) Control of the Orientational Order and Nonlinear Optical Response of the "Push-Pull" Chromophore RuPZn via Specific Incorporation into Densely-Packed Monolayer Ensembles of an Amphiphilic 4-Helix Bundle Peptide: Second Harmonic Generation at High Chromophore Densities. J. Am. Chem. Soc. 132 (28): 9693–9700.
http://pubs.acs.org/Volume=132 (28)
Krishnan, V., Tronin, A., Strzalka, J., Fry, H.C., Therien, M.J. and Blasie, J.K. (2010) Control of the Orientational Order and Nonlinear Optical Response of the “Push-Pull” Chromophore RuPZn via Specific Incorporation into Densely-Packed Monolayer Ensembles of an Amphiphilic 4-Helix Bundle Peptide: Characterization of the Cofactor-Peptide Complex in Monolayer Ensembles. J. Am. Chem. Soc. 132(32):11083-11092.
http://pubs.acs.org/Volume=132 (32)
Krishnan, V., Strzalka, J., Liu, J., Liu, C., Kuzmenko, I., Gog, T. and Blasie, J.K. (2010) Interferometric Enhancement of X-ray Reflectivity from Unperturbed Langmuir Monolayers of Amphiphiles at the Liquid-Gas Interface. Phys. Rev. E 81: 021604-1-10.
Zou, H. Liu, J. and Blasie, J.K. (2009) Mechanism of interaction between the volatile anesthetic halothane and a model ion channel protein: III. Molecular dynamics simulation incorporating a cyano-phenylalanine spectroscopic probe. Biophys. J. 96(10) pp. 4188 – 4199.
Liu, J., Strzalka, J., Tronin, A., Johansson, J.S. and Blasie, J.K. (2009) Mechanism of interaction between the volatile anesthetic halothane and a model ion channel protein: II. Fluorescence & vibrational spectroscopy employing a cyano-phenylalanine probe. Biophys. J. 96(10) pp. 4176 – 4187.
Strzalka, J., Liu, J., Tronin, A., Johansson, J.S. and Blasie, J.K. (2009) Mechanism of interaction between the volatile anesthetic halothane and a model ion channel protein: I. Structural investigations via x-ray reflectivity from Langmuir monolayers. Biophys. J. 96(10) pp. 4164 – 4175.
Tronin, A., Krishnan, V., Strzalka, J., Kuzmenko, I., Gog, T., Fry, C., Therien, M.J. and Blasie, J.K. (2009) Portable UV-VIS spectrometer for measuring absorbance and dichroism of Langmuir monolayers. Rev. Sci. Instru. 80(3): 033102-1-7.
Zou, H., Therein, M.J. and Blasie, J.K. Structure & Dynamics of an Extended Conjugated NLO Chromophore within an Amphiphilic 4-Helix Bundle Peptide by Molecular Dynamics Simulation. Submitted to J. Phys. Chem. B.
McAllister, K.A., Zou, H., Cockran, F.V., Bender, G.M., Senes, A., Fry, C.F., Nanda, V., Keenan, P.A., Lear, J.D., Therien, M.J., Blasie, J.K, and DeGrado, W.F. Using α-Helical Coiled-Coils to Design Nanostructured Metalloporphyrin Arrays. Submitted to Angewandte Chemie.
Bender, G.M., Lehmann, A., Zou, H., Cheng, H., Fry, H.C., Engel, D., Therien, M.J., Blasie, J.K., Roder, H., Saven, J.G. and DeGrado, W.F. De Novo Design of a Single Chain Diphenylporphyrin Metalloprotein. J. Am. Chem. Soc. 129(35).
http://pubs.acs.org/Volume=129
Zou, H., Strzalka, J. Xu, T., Tronin, A. and Blasie, J.K. (2007) 3-D Structure and Dynamics via Molecular Dynamics Simulation of a de novo Designed, Amphiphilic Heme Protein Maquette at Soft Interfaces. Phys. Chem. B 111: 1823-1833.
Strzalka, J., Xu, T., Tronin, A., Wu, S.P., Miloradovic, I., Kuzmenko, I. Gog, T., Therien, M.J., and Blasie, J.K. (2006) Structural Studies of Amphiphilic 4-helix Bundle Peptides Incorporating Designed Extended Chromophores for Nonlinear Optical Biomolecular Materials. Nano Lett. 6(11): 2395-2405.
Xu, T., Wu, S.P., Miloradovic, I., Therien, M.J., and Blasie, J.K. (2006) Incorporation of Designed Extended Chromophores into Amphiphilic 4-helix Bundle Peptides for Nonlinear Optical Biomolecular Materials. Nano Lett. 6(11):
http://pubs.acs.org/Volume=6(2387-2394)
Nordgren, C.E., Strzalka, J.W. and Blasie, J.K (2005) Structure of α-Helical Bundle Peptides Vectorially-Oriented at Soft Interfaces via Molecular Dynamics Simulations and X-ray/Neutron Scattering. Submitted to Langmuir.
Churbanova, I., Tronin, A., Strzalka, J.W., Gog, T., Kuzmenko, I., Johansson, J.S. and Blasie, J.K. (2006) Monolayers of a Model Anesthetic-Binding Membrane Protein: Formation, Characterization and Halothane-Binding Affinity. Biophys. J. 90: 3255-3266.
http://www.cell.com/biophysj/Volume=90
Tronin, A., Xu, T. and Blasie, J.K. (2005) In situ Determination of Orientational Distributions in Langmuir Monolayers by Total Internal Reflection Fluorescence. Langmuir 21: 7760-7767.
Discher, B.M., Noy, D., Strazalka, J, Ye, S., Moser, C.C., Lear, J.D., Blasie, J.K. and Dutton, P.L. (2005) Design of Amphiphilic Protein Maquettes: Controlling Assembly, Membrane Insertion, and Cofactor Interactions. Biochemistry 44:12329-12343.
Ye, S., Discher, B.M., Strzalka, J.W., Xu, T., Wu, S.P., Noy, D., Kuzmenko, I., Gog, T., Therien, M.J., Dutton, P.L. and Blasie, J.K. (2005) Amphiphilic 4-Helix Bundles Designed for Light-Induced Electron Transfer Across Soft Interfaces. Nano Lett. 5(9):1658-1667.
Ye, S., Strzalka, J., Churbanova, I.Y., Zheng, S,, Johansson, J.S. and Blasie JK. (2004) A Model Membrane Protein for Binding Volatile Anesthetics. Biophys. J. 87: 4065-4074.
http://www.cell.com/biophysj/Volume=87
Strzalka, J. DiMasi, E., Kuzmenko, I., Gog, T. and Blasie, J.K. (2004) Resonant X-ray Reflectivity from a Bromine-Labeled Fatty-Acid Langmuir Monolayer. Phys. Rev. E 70: 051603-1-5.
http://pre.aps.org/abstract/PRE/v70/i5/e051603
Strzalka, J., Kneller, L.R., Gibney, B.R., Satija, S., Majkrzak, C.F. and Blasie, J.K. (2004) Specular Neutron Reflectivity and Structure of Artificial Protein Maquettes Vectorially Oriented at Interfaces. Phys. Rev. E. E 70: 061905-1-10.
http://pre.aps.org/abstract/PRE/v70/i6/e061905
Ye, S., Discher, B.M., Strzalka, J.W., Noy, D., Zheng, S., Dutton, P.L. and Blasie, J.K. (2004) Amphiphilic 4-Helix Bundles Designed for BioMolecular Materials Applications. Langmuir 20(14): 5897-5904.
Blasie, J.K., Strzalka, J. and Zheng, S. (2003) Solution to the Phase Problem for Specular X-ray & Neutron Reflectivity from Thin Films on Liquid Surfaces. Phys. Rev. B 67: 224201-1--224201-8.
Ye, S., Strzalka, J., Chen, X., Moser, C.C., Dutton, P.L. and Blasie, J.K. (2003) Assembly of a Vectorially-Oriented Four-Helix Bundle at the Air/Water Interface via Directed Electrostatic Interactions. Langmuir 19(5): 1515-1521.
Zheng, S., Strzalka, J., Jones, D.H., Opella, S.J. and Blasie, J.K. (2003) Comparative Structural Studies of Vpu Peptides in Phospholipid Monolayers by X-ray Scattering. Biophys. J. 84(4): 2393-2415.
Lopez, C. F., Montal, M., Blasie, J.K., Klein, M.L. and Moore, P.B. (2002) Molecular Dynamics Investigation of Membrane-Bound Bundles of the Channel Forming Transmembrane Domain of Viral Protein U from the Human Immunodeficiency Virus HIV. Biophys. J. 83(3): 1259-1267.
Nordgren, E., Tobias, D.J., Klein, M.L. and Blasie, J.K. (2002) Molecular Dynamics Simulations of a Hydrated Protein Vectorially-Oriented at Hydrophobic vs. Hydrophilic Soft Surfaces. Biophys. J. 83(6): 2906-2917.
http://www.cell.com/biophysj/Volume=83
Tronin, A., Edwards, A.M., Wright, W.W., Vanderkooi, J.M. and Blasie, J.K. (2002) Orientational Distributions for Cytochrome c on Polar & Nonpolar Soft Surfaces by Polarized Total Internal Reflection Fluorescence. Biophys. J. 82: 996-1003.
http://www.cell.com/biophysj/Volume=82
Haas, A.S., Pilloud, D.L., Reddy, K.S., Babcock, G.T., Moser, C.C., Blasie, J.K. and Dutton, P.L. Cytochrome c and Cytochrome c Oxidase: Monolayer Assemblies and Catalysis. (2001) J. Phys. Chem. B 105(45): 11351-11362.
http://pubs.acs.org/Volume=105
Tronin, A. and Blasie, J.K. Variable Acquisition Angle Total Internal Reflection Fluorescence – a New Technique for Orientation Distribution Studies of Ultrathin Films. (2001) Langmuir 17(No. 12): 3696-3703.
Tronin, A., Strzalka, J., Chen, X., Dutton, P.L., Ocko, B.M. and Blasie, J.K. (2001) Orientational Distributions of the Di-α-Helical Synthetic Peptide ZnPPIX-BBC16 by X-ray Reflectivity and Polarized Epifluorescence. Langmuir 17(10): 3061-3066.
Zheng, S., Strzalka, J., Ma, C., Opella, S.J., Ocko, B.M. and Blasie, J.K. (2001) Structural Studies of the HIV-1 Accessory Protein Vpu in Langmuir Monolayers: Synchrotron X-ray Reflectivity. Biophys. J. 80(4): 1837-1850.
http://www.cell.com/biophysj/Vol80
Kneller, L.R. , Edwards, A.M., Majkrzak, C.F., Berk, N.F., Krueger, S. and Blasie, J.K. (2001) Hydration State of a Single Cytochrome c Monolayer Vectorially-Oriented at a Soft Interface Investigated via Neutron Interferometry. Biophys. J. 80(5): 2248-2261.
http://www.cell.com/biophysj/s?searchTerms=&searchAuthor=&searchVol80
Strzalka, J., Chen, X., Dutton, P.L. and Blasie, J.K. (2001) X-ray Scattering Studies of Maquette Peptide Monolayers II: Interferometry at the Solid/Vapor Interface. Langmuir 17(4): 1193-1199.
Tronin, A., Strzalka, J., Chen, X., Dutton, P.L. and Blasie, J.K. (2000) Determination of the Porphyrin Orientation Distribution in Langmuir Monolayers by Polarized Epifluorescence. Langmuir 16(25): 9878-9886.
Strzalka, J., Chen, X., Dutton, P.L., Ocko, B. M. and Blasie, J.K. (2000) X-ray Scattering Studies of Maquette Peptide Monolayers I: Reflectivity and GID at the Air/Water Interface. Langmuir 16(26): 10404-10418.
A.M. Edwards, K. Zhang, C.E. Nordgren and J.K. Blasie. (2000) Heme Structure & Orientation in Single Monolayers of Cytochrome c on Polar & Nonpolar Soft Surfaces. Biophys. J. 79: 3105-3117.
http://www.cell.com/biophysj/Vol 79
Blasie, J.K. and Timmins, P. (1999) Neutron Scattering in Structural Biology & BioMolecular Materials in Neutron Scattering in Materials Research, eds. T. Mason and A. Taylor, MRS Bulletin 24(12): 40-47.

